00019334142026Q1False12/31446xbrli:sharesiso4217:USDiso4217:USDxbrli:sharesmlys:segmentxbrli:pure00019334142026-01-012026-03-3100019334142026-04-2700019334142026-03-3100019334142025-12-3100019334142025-01-012025-03-310001933414us-gaap:CommonStockMember2024-12-310001933414us-gaap:AdditionalPaidInCapitalMember2024-12-310001933414us-gaap:RetainedEarningsMember2024-12-3100019334142024-12-310001933414mlys:PublicOfferingMember2025-01-012025-03-310001933414mlys:PublicOfferingMemberus-gaap:CommonStockMember2025-01-012025-03-310001933414mlys:PublicOfferingMemberus-gaap:AdditionalPaidInCapitalMember2025-01-012025-03-310001933414us-gaap:CommonStockMember2025-01-012025-03-310001933414us-gaap:AdditionalPaidInCapitalMember2025-01-012025-03-310001933414us-gaap:RetainedEarningsMember2025-01-012025-03-310001933414us-gaap:CommonStockMember2025-03-310001933414us-gaap:AdditionalPaidInCapitalMember2025-03-310001933414us-gaap:RetainedEarningsMember2025-03-3100019334142025-03-310001933414us-gaap:CommonStockMember2025-12-310001933414us-gaap:AdditionalPaidInCapitalMember2025-12-310001933414us-gaap:RetainedEarningsMember2025-12-310001933414mlys:AtTheMarketMember2026-01-012026-03-310001933414mlys:AtTheMarketMemberus-gaap:CommonStockMember2026-01-012026-03-310001933414mlys:AtTheMarketMemberus-gaap:AdditionalPaidInCapitalMember2026-01-012026-03-310001933414us-gaap:CommonStockMember2026-01-012026-03-310001933414us-gaap:AdditionalPaidInCapitalMember2026-01-012026-03-310001933414us-gaap:RetainedEarningsMember2026-01-012026-03-310001933414us-gaap:CommonStockMember2026-03-310001933414us-gaap:AdditionalPaidInCapitalMember2026-03-310001933414us-gaap:RetainedEarningsMember2026-03-310001933414mlys:AtTheMarketMember2025-01-012025-03-310001933414mlys:PublicOfferingMember2026-01-012026-03-3100019334142023-02-142026-03-310001933414us-gaap:EmployeeStockOptionMember2026-01-012026-03-310001933414us-gaap:EmployeeStockOptionMember2025-01-012025-03-310001933414us-gaap:RestrictedStockUnitsRSUMember2026-01-012026-03-310001933414us-gaap:RestrictedStockUnitsRSUMember2025-01-012025-03-310001933414us-gaap:RestrictedStockMember2026-01-012026-03-310001933414us-gaap:RestrictedStockMember2025-01-012025-03-310001933414us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2026-03-310001933414us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2025-12-310001933414us-gaap:InvestmentsMember2026-03-310001933414us-gaap:CashAndCashEquivalentsMember2025-12-310001933414us-gaap:InvestmentsMember2025-12-310001933414mlys:TanabeMemberus-gaap:CollaborativeArrangementTransactionWithPartyToCollaborativeArrangementMember2020-07-310001933414us-gaap:CollaborativeArrangementTransactionWithPartyToCollaborativeArrangementMembermlys:TanabeMembersrt:MaximumMember2020-07-310001933414mlys:TanabeMemberus-gaap:CollaborativeArrangementTransactionWithPartyToCollaborativeArrangementMember2025-03-310001933414mlys:TanabeMemberus-gaap:CollaborativeArrangementTransactionWithPartyToCollaborativeArrangementMember2026-03-310001933414us-gaap:CollaborativeArrangementTransactionWithPartyToCollaborativeArrangementMembermlys:TanabeMembersrt:MinimumMember2020-07-310001933414us-gaap:EmployeeStockOptionMember2026-03-310001933414mlys:EquityIncentivePlan2023Member2026-03-310001933414us-gaap:EmployeeStockMember2026-03-310001933414us-gaap:RestrictedStockUnitsRSUMember2026-03-310001933414mlys:PreFundedWarrantMember2026-03-310001933414mlys:EquityIncentivePlan2025Member2026-03-310001933414srt:MaximumMembermlys:AtTheMarketMember2025-11-100001933414mlys:AtTheMarketMember2025-04-012026-03-310001933414mlys:AtTheMarketMember2026-03-310001933414mlys:PriorATMAgreementMember2024-03-212025-11-090001933414mlys:PriorATMAgreementMember2025-11-090001933414mlys:A2023IncentiveAwardPlanMember2026-03-310001933414mlys:A2023IncentiveAwardPlanMemberus-gaap:RestrictedStockMember2026-03-310001933414mlys:A2023IncentiveAwardPlanMemberus-gaap:RestrictedStockUnitsRSUMember2026-03-310001933414mlys:A2025IncentiveAwardPlanMember2026-03-310001933414mlys:A2025IncentiveAwardPlanMemberus-gaap:RestrictedStockMember2026-03-310001933414mlys:A2025IncentiveAwardPlanMemberus-gaap:RestrictedStockUnitsRSUMember2026-03-310001933414mlys:A2020EquityIncentivePlanMember2026-03-310001933414mlys:A2020EquityIncentivePlanMemberus-gaap:RestrictedStockMember2026-03-310001933414mlys:A2020EquityIncentivePlanMemberus-gaap:RestrictedStockUnitsRSUMember2026-03-310001933414us-gaap:RestrictedStockMember2026-03-310001933414mlys:A2023EmployeeStockPurchasePlanMemberus-gaap:EmployeeStockMember2026-03-310001933414mlys:A2023EmployeeStockPurchasePlanMemberus-gaap:EmployeeStockMember2026-01-012026-03-310001933414us-gaap:ResearchAndDevelopmentExpenseMember2026-01-012026-03-310001933414us-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-03-310001933414us-gaap:GeneralAndAdministrativeExpenseMember2026-01-012026-03-310001933414us-gaap:GeneralAndAdministrativeExpenseMember2025-01-012025-03-310001933414mlys:CatalysPacificFundLPMember2026-01-012026-03-310001933414mlys:CatalysPacificFundLPMember2026-03-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
(Mark One)
| | | | | |
| x | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended March 31, 2026
OR
| | | | | |
¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from ______ to ______
Commission file number 001-41614
| | | | | | | | |
| | |
| | |
MINERALYS THERAPEUTICS, INC. |
| | |
| | |
(Exact name of registrant as specified in its charter)
| | | | | |
Delaware | 84-1966887 |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| |
150 N. Radnor Chester Road, Suite F200, Radnor, PA 19087 |
(Address, including zip code, of principal executive offices) |
| |
888-378-6240 |
(Registrant's telephone number, including area code) |
(Former name, former address and former fiscal year, if changed since last report): NA
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
Common stock, $0.0001 par value per share | MLYS | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports); and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | | | | |
Large accelerated filer | o | | Accelerated filer | o |
Non-accelerated filer | x | | Smaller reporting company | x |
| | | Emerging growth company | x |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes o No x
As of April 27, 2026, there were 82,498,101 shares of the registrant’s common stock outstanding.
Mineralys Therapeutics, Inc.
Table of Contents
Part I - Financial Information
Item 1. Financial Statements
Mineralys Therapeutics, Inc.
Condensed Balance Sheets
(in thousands, except share and per share amounts)
| | | | | | | | | | | |
| |
| | | |
| March 31, | | December 31, |
| 2026 | | 2025 |
| (unaudited) | | |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 112,779 | | | $ | 172,921 | |
| Investments | 533,281 | | | 483,714 | |
| Prepaid and other current assets | 6,410 | | | 4,751 | |
| Total current assets | 652,470 | | | 661,386 | |
| | | |
| Property and equipment, net | 32 | | | 38 | |
| Other assets | 360 | | | 382 | |
| Total assets | $ | 652,862 | | | $ | 661,806 | |
| | | |
| Liabilities and stockholders’ equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 1,116 | | | $ | 2,017 | |
| Accrued liabilities | 13,602 | | | 13,096 | |
| | | |
| Total current liabilities | 14,718 | | | 15,113 | |
| Commitments and contingencies (Note 4) | | | |
| | | |
| | | |
| Stockholders’ equity: | | | |
Common stock, $0.0001 par value; 500,000,000 shares authorized as of March 31, 2026 and December 31, 2025; 82,454,950 and 81,536,557 shares issued and outstanding as of March 31, 2026 and December 31, 2025, respectively | 8 | | | 8 | |
| Additional paid-in capital | 1,134,644 | | | 1,103,854 | |
| Accumulated deficit | (496,508) | | | (457,169) | |
| Total stockholders’ equity | 638,144 | | | 646,693 | |
| Total liabilities and stockholders’ equity | $ | 652,862 | | | $ | 661,806 | |
The accompanying notes are an integral part of these condensed financial statements.
Mineralys Therapeutics, Inc.
Condensed Statements of Operations
(in thousands, except share and per share data)
(unaudited)
| | | | | | | | | | | | | | | |
| | | Three Months Ended |
| | | March 31, |
| | | | | 2026 | | 2025 |
| Operating expenses: | | | | | | | |
| Research and development | | | | | $ | 24,365 | | | $ | 37,879 | |
| General and administrative | | | | | 20,975 | | | 6,568 | |
| Total operating expenses | | | | | 45,340 | | | 44,447 | |
| Loss from operations | | | | | (45,340) | | | (44,447) | |
| | | | | | | |
| Interest income, net | | | | | 5,996 | | | 2,239 | |
| | | | | | | |
| Other income (expense) | | | | | 5 | | | (3) | |
| Total other income, net | | | | | 6,001 | | | 2,236 | |
| Net loss | | | | | $ | (39,339) | | | $ | (42,211) | |
| Net loss per share attributable to common stockholders, basic and diluted | | | | | $ | (0.47) | | | $ | (0.79) | |
| Weighted-average shares used in computing net loss per share attributable to common stockholders, basic and diluted | | | | | 82,834,752 | | | 53,163,551 | |
The accompanying notes are an integral part of these condensed financial statements.
Mineralys Therapeutics, Inc.
Condensed Statements of Stockholders’ Equity
(in thousands, except share data)
(unaudited)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | Common Stock | | Additional
Paid-In Capital | | Accumulated
Deficit | | Total
Stockholders’ Equity |
| | | | | | | | | | Shares | | Amount | |
| Balance as of December 31, 2024 | | | | | | | | | | 49,821,915 | | | $ | 5 | | | $ | 493,770 | | | $ | (302,518) | | | $ | 191,257 | |
Issuance of common stock in public offering, net of offering costs of $12,509 | | | | | | | | | | 14,907,406 | | | 1 | | | 188,740 | | | — | | | 188,741 | |
| Issuance of common stock from stock option exercises | | | | | | | | | | 146,907 | | | — | | | 123 | | | — | | | 123 | |
| | | | | | | | | | | | | | | | | | |
| Stock-based compensation | | | | | | | | | | — | | | — | | | 3,645 | | | — | | | 3,645 | |
| Net loss | | | | | | | | | | — | | | — | | | — | | | (42,211) | | | (42,211) | |
| Balance as of March 31, 2025 | | | | | | | | | | 64,876,228 | | | $ | 6 | | | $ | 686,278 | | | $ | (344,729) | | | $ | 341,555 | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Common Stock | | Additional
Paid-In Capital | | Accumulated
Deficit | | Total
Stockholders’ Equity |
| | | | | | | | | | Shares | | Amount | |
| Balance as of December 31, 2025 | | | | | | | | | | 81,536,557 | | | $ | 8 | | | $ | 1,103,854 | | | $ | (457,169) | | | $ | 646,693 | |
Issuance of common stock pursuant to ATM Agreement, net of issuance costs and shelf offering costs of $82 | | | | | | | | | | 568,320 | | | — | | | 20,187 | | | — | | | 20,187 | |
| Issuance of common stock from stock option exercises | | | | | | | | | | 350,073 | | | — | | | 4,055 | | | — | | | 4,055 | |
| | | | | | | | | | | | | | | | | | |
| Stock-based compensation | | | | | | | | | | — | | | — | | | 6,548 | | | — | | | 6,548 | |
| Net loss | | | | | | | | | | — | | | — | | | — | | | (39,339) | | | (39,339) | |
| Balance as of March 31, 2026 | | | | | | | | | | 82,454,950 | | | $ | 8 | | | $ | 1,134,644 | | | $ | (496,508) | | | $ | 638,144 | |
The accompanying notes are an integral part of these condensed financial statements.
Mineralys Therapeutics, Inc.
Condensed Statements of Cash Flows
(in thousands)
(unaudited)
| | | | | | | | | | | |
| Three Months Ended |
| March 31, |
| 2026 | | 2025 |
| CASH FLOWS FROM OPERATING ACTIVITIES: | | | |
| Net loss | $ | (39,339) | | | $ | (42,211) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | |
| Stock-based compensation | 6,548 | | | 3,645 | |
| Accretion of discount on held-to-maturity securities | (4,625) | | | (1,046) | |
| Depreciation and amortization | 6 | | | 15 | |
| | | |
| | | |
| Changes in operating assets and liabilities: | | | |
| Accrued interest receivable | 289 | | | 52 | |
Prepaid, other current assets, and other assets | (1,948) | | | (4,396) | |
| Accounts payable and accrued liabilities | (402) | | | (1,546) | |
| Net cash used in operating activities | (39,471) | | | (45,487) | |
| | | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | | | |
| Purchases of marketable securities | (347,443) | | | (127,876) | |
| Maturities of marketable securities | 302,500 | | | 35,000 | |
| | | |
| | | |
| Net cash used in investing activities | (44,943) | | | (92,876) | |
| | | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | | | |
| | | |
Proceeds from issuance of common stock pursuant to ATM Agreement, net of ATM issuance costs | 20,218 | | | — | |
| | | |
| Proceeds from stock option exercises | 4,055 | | | 123 | |
| | | |
| | | |
| | | |
| | | |
| | | |
Payment of shelf offering costs | (1) | | | (10) | |
| Proceeds from issuance of common stock in public offering, net of offering costs | — | | | 189,166 | |
| | | |
| | | |
| Net cash provided by financing activities | 24,272 | | | 189,279 | |
| | | |
| Net increase (decrease) in cash and cash equivalents | (60,142) | | | 50,916 | |
| Cash and cash equivalents - beginning | 172,921 | | | 114,091 | |
Cash and cash equivalents - ending(1) | $ | 112,779 | | | $ | 165,007 | |
| | | |
| Supplemental Disclosure of Non-Cash Financing Activities: | | | |
| | | |
| Public offering and shelf offering costs included in accounts payable and accrued liabilities | $ | 7 | | | $ | 289 | |
| | | |
(1)Cash and cash equivalents as of March 31, 2026 exclude investments of $533.3 million. Cash, cash equivalents, and investments amounted to $646.1 million as of March 31, 2026.
The accompanying notes are an integral part of these condensed financial statements.
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
Note 1. Nature of Business
Mineralys Therapeutics, Inc. (the Company) is a biopharmaceutical company focused on developing medicines to target diseases driven by dysregulated aldosterone. The Company’s product candidate, lorundrostat, is a proprietary, orally administered, aldosterone synthase inhibitor that the Company is developing for the treatment of cardiorenal conditions affected by dysregulated aldosterone, including hypertension and related comorbidities, such as chronic kidney disease and obstructive sleep apnea. The Company has completed two pivotal clinical trials of lorundrostat for hypertension, one Phase 2 clinical trial in hypertensive participants with chronic kidney disease, and one Phase 2 clinical trial in hypertensive participants with obstructive sleep apnea. The Company submitted a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) in December 2025 for lorundrostat for the treatment of hypertension in combination with other antihypertensive drugs. The FDA accepted the NDA submission and provided the Company with a Prescription Drug User Fee Act target date of December 22, 2026 for lorundrostat. The Company was incorporated as a Delaware corporation in May 2019, and it is headquartered in Radnor, Pennsylvania. The Company’s operations to date have been limited to business planning, raising capital, in-licensing, conducting preclinical and clinical trials, and performing research and development and other activities.
Liquidity and Capital Resources
Since its inception, the Company has not generated any revenue from product sales or other sources and has incurred significant operating losses and negative cash flows from operations. The Company’s primary uses of cash to date have been to fund research and development activities, business planning, establishing and maintaining the Company’s intellectual property portfolio, hiring personnel, raising capital, and providing general and administrative support for these operations. As of March 31, 2026, the Company had an accumulated deficit of $496.5 million and cash, cash equivalents, and investments of $646.1 million. For the three months ended March 31, 2026, the Company had a net loss of $39.3 million and net cash used in operating activities of $39.5 million.
Since inception, the Company has funded its operations by raising aggregate gross proceeds of approximately $1.1 billion from the sale of the Company’s common stock, convertible preferred stock, pre-funded warrants, and convertible notes. The Company has a limited operating history, and the sales and income potential of its business is unproven. The Company expects to continue to incur substantial losses for the foreseeable future as a result of the Company’s research and development and other activities. Additional funding will be required to continue with the Company’s planned research and development and commercial launch activities. The Company expects to finance its operations through equity offerings, debt financings, and other capital sources, including potential strategic collaborations, licensing, and other similar arrangements. The Company believes that its cash, cash equivalents, and investments as of March 31, 2026 will be sufficient to allow the Company to fund operations for at least twelve months from the issuance date of these condensed financial statements.
Note 2. Summary of Significant Accounting Policies
Basis of Presentation
The unaudited condensed financial statements have been prepared in accordance with accounting principles generally accepted in the United States (U.S. GAAP) and applicable rules and regulations of the U.S. Securities and Exchange Commission (SEC) for interim reporting. As permitted under those rules and regulations, certain footnotes or other financial information normally included in financial statements prepared in accordance with U.S. GAAP have been condensed or omitted. These condensed financial statements have been prepared on the same basis as the annual financial statements and, in the opinion of management, reflect all adjustments, consisting only of normal recurring adjustments, which are necessary for a fair presentation of the Company’s financial information. These unaudited condensed financial statements should be read in
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
conjunction with the audited financial statements and related notes in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025.
Any reference in these notes to applicable guidance is meant to refer to authoritative U.S. GAAP as found in the Accounting Standards Codification (ASC) and Accounting Standards Updates (ASU) of the Financial Accounting Standards Board (FASB).
Segment Information
The Company operates in one operating segment for the purposes of assessing performance, making operating decisions, and allocating Company resources. The Company’s chief operating decision maker (CODM) is its chief executive officer, who considers net loss to evaluate overall expenses associated with conducting research and development activities, which includes evaluating the progress of ongoing clinical trials and the planning and execution of current and future research and development and other activities. Further, the CODM reviews and utilizes functional expenses (research and development and general and administrative) as reported in the statements of operations to manage the Company’s operations. Other segment items included in net loss are interest income, net and other income (expense). These measures of performance, significant expenses, and other items are each reflected in the condensed statements of operations. The accounting policies of the segment are the same as those described below. The measure of segment assets is reported on the condensed balance sheets as total assets. All assets are held in the United States.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the condensed financial statements and the reported amounts of income and expenses during the reporting period. Actual results could differ from those estimates. Estimates have been used in the following areas, among others: research and development accruals and income taxes.
Cash and Cash Equivalents
All highly liquid investments that have maturities of 90 days or less at the date of purchase are classified as cash equivalents. The Company’s cash and cash equivalents balances as of March 31, 2026 and December 31, 2025 include cash balances and amounts held primarily in interest-bearing money market accounts and U.S. Treasury bills. As of March 31, 2026 and December 31, 2025, the Company did not have any restricted cash balances. The following table provides a reconciliation of cash and cash equivalents as reported in the condensed statements of cash flows to the condensed balance sheets (in thousands):
| | | | | | | | | | | |
| |
| | | |
| March 31, | | December 31, |
| 2026 | | 2025 |
| Cash | $ | 1,150 | | | $ | 1,030 | |
| Cash equivalents | 111,629 | | | 171,891 | |
| Total cash and cash equivalents | $ | 112,779 | | | $ | 172,921 | |
| | | |
| | | |
Concentration of Credit Risk
The Company has no significant off-balance sheet concentrations of credit risk, such as foreign currency exchange contracts, option contracts, or other hedging arrangements. Financial instruments that potentially subject the Company to concentrations of credit risk primarily consist of cash balances in several accounts with three financial institutions that, from time to time, are in excess of federally insured limits.
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
Fair Value Measurements
The Company is required to disclose information on all assets and liabilities reported at fair value that enables an assessment of the inputs used in determining the reported fair values. ASC Topic 820, Fair Value Measurement, establishes a hierarchy of inputs used in measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that the observable inputs be used when available.
Observable inputs are inputs that market participants would use in pricing the asset or liability based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the asset or liability and are developed based on the best information available in the circumstances. The fair value hierarchy applies only to the valuation inputs used in determining the reported fair value and is not a measure of the investment credit quality. The three levels of the fair value hierarchy are described below:
•Level 1 – quoted prices in active markets for identical assets and liabilities
•Level 2 – other significant observable inputs (including quoted prices for similar assets and liabilities, interest rates, credit risk, etc.)
•Level 3 – significant unobservable inputs (including the Company’s own assumptions in determining the fair value of assets and liabilities)
For certain financial instruments, including cash and cash equivalents, prepaid expenses, accounts payable, and certain accrued liabilities, the recorded amount approximates estimated fair value due to their relatively short maturity period. Refer to Note 3. “Fair Value of Financial Instruments” for additional details of the Company’s financial instruments.
Investments
The Company generally invests its excess cash in money market funds and investment-grade short- and long-term fixed-income debt securities, such as U.S. Treasury bills. Such investments are included in cash and cash equivalents and investments in the condensed balance sheets. The Company determines the appropriate classification of securities at the time of purchase and re-evaluates such designation as of each balance sheet date. Securities are classified as held-to-maturity when the Company has the positive intent and ability to hold the securities to maturity. Held-to-maturity securities are carried at amortized cost, adjusted for the accretion of discounts using the interest method.
The Company invested in marketable securities during the three months ended March 31, 2026 and 2025, and no impairment charges were recorded. For held-to-maturity investments, the Company periodically reviews each individual security position that has an unrealized loss, or impairment, to determine if that impairment is other-than-temporary. If the Company believes an impairment of a security position is other than temporary, based on available quantitative and qualitative information as of the report date, the loss will be recognized within total other income, net in the Company’s condensed statements of operations, and a new cost basis in the investment will be established.
Deferred Offering Costs
The Company capitalizes certain legal, professional, accounting, and other third-party fees that are directly associated with in-process equity issuances as deferred offering costs until such equity issuances are consummated. After consummation of the equity issuance, these costs are recorded as a reduction in the capitalized amount associated with the equity issuance. Should the equity issuance be abandoned, the deferred offering costs are expensed immediately as a charge to operating expenses in the condensed statements of operations. Deferred offering costs as of each of March 31, 2026 and December 31, 2025 were $0.2 million. Such costs are classified in other assets on the condensed balance sheets.
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
Stock-Based Compensation
The Company accounts for its stock-based compensation awards in accordance with ASC Topic 718, Compensation – Stock Compensation (ASC 718). ASC 718 requires share-based payment arrangements to be recognized in the condensed statements of operations based on their grant date fair values. The Company’s stock-based awards are subject only to service-based vesting conditions. The Company measures restricted common stock awards using the difference, if any, between the purchase price per share of the award and the fair value of the Company’s common stock at the date of the grant or modification. Restricted stock unit awards are measured at the grant date fair value based on the closing price of the Company’s common stock on the date of the grant. The Company estimates the fair value of its stock option awards using the Black-Scholes option pricing model, which requires the input of assumptions, including (i) the expected stock price volatility, (ii) the calculation of the expected term of the award, (iii) the risk-free interest rate, and (iv) expected dividends.
Volatility — Due to the Company’s limited operating history and a lack of company-specific historical and implied volatility data, the Company has based its estimate of expected volatility on the historical volatility of a group of similar publicly-traded companies. The Company believes that the companies in the group were most representative of the Company and had characteristics similar to its own, including stage of product development, a focus on the life sciences industry, and other economic and industry characteristics.
Expected Term — The Company uses the simplified method to calculate the expected term, as it does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate the expected term for options granted, and utilizes the contractual term for options granted.
Risk-Free Interest Rate — The risk-free interest rate is based on a treasury instrument whose term is consistent with the expected life of the stock options.
Expected Dividends — To date, the Company has not issued any dividends and does not expect to issue dividends over the life of the options and therefore has estimated the dividend yield to be zero.
Subsequent to the closing of the Company’s initial public offering of its common stock in February 2023, the Company determines the fair market value of its common stock using the closing price of its common stock as reported on the Nasdaq Global Select Market.
Compensation expense related to time-based awards, including restricted stock units, is recognized on a straight-line basis by recognizing the grant date fair value over the requisite service period, which is generally the vesting term. Management evaluates its award grants and modifications and will adjust the fair value if any are determined to be spring-loaded. The Company accounts for forfeitures as they occur.
Net Loss Per Share
The Company’s basic net loss per share attributable to common stockholders is calculated by dividing the net loss attributable to common stockholders by the weighted-average number of shares of common stock outstanding for the period. The diluted net loss per share attributable to common stockholders is computed by giving effect to all potential common stock equivalents outstanding for the period, determined using the treasury stock method. For purposes of this calculation, unvested restricted stock awards, restricted stock units, and stock options to purchase common stock are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share attributable to common stockholders as their effect is anti-dilutive. The weighted-average number of common shares used in the basic and diluted net loss per share attributable to common stockholders calculations includes the weighted-average pre-funded warrants outstanding during the period, as they are exercisable at any time for nominal cash consideration. The following table sets forth the
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
potential common shares excluded from the calculation of net loss per share attributable to common stockholders because their inclusion would be anti-dilutive:
| | | | | | | | | | | |
| Three Months Ended |
| March 31, |
| 2026 | | 2025 |
| Outstanding options | 7,420,692 | | | 6,778,317 | |
| Unvested restricted stock units | 702,948 | | | — | |
| Unvested restricted stock awards | 64,714 | | | 309,028 | |
| Total | 8,188,354 | | | 7,087,345 | |
Recently Issued Accounting Pronouncements
From time to time, new accounting pronouncements are issued by the FASB or other standard-setting bodies and adopted by the Company as of the specified effective date. Unless otherwise discussed, the Company believes the impact of recently issued standards that are not yet effective will not have a material impact on its financial position or results of operations upon adoption.
In November 2024, the FASB issued ASU 2024-03, Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses (ASU 2024-03), and in January 2025, the FASB issued ASU 2025-01, Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Clarifying the Effective Date, which clarified the effective date of ASU 2024-03. ASU 2024-03 will require the Company to disclose additional information about specific expense categories in the notes to the financial statements on an annual and interim basis. ASU 2024-03 is effective for the Company's fiscal year 2027 and may be adopted on a prospective or retrospective basis. Early adoption is permitted. The Company is evaluating the impact of ASU 2024-03 on its disclosures. While the Company expects that additional disaggregated disclosures will be required upon adoption, the guidance will not affect the recognition or measurement of expenses.
Note 3. Fair Value of Financial Instruments
The following table presents financial instruments measured at fair value on a recurring basis based on the fair value hierarchy as of March 31, 2026 and December 31, 2025 (in thousands):
| | | | | | | | | | | |
| |
| | | |
| March 31, | | December 31, |
| 2026 | | 2025 |
| Level 1 |
| Assets | | | |
| Cash equivalents | | | |
| Money market funds | $ | 111,629 | | | $ | 161,943 | |
| | | |
| | | |
There were no transfers within the fair value hierarchy during the periods presented.
The following methods and assumptions were used by the Company in estimating the fair values of each class of financial instrument disclosed herein:
Money Market Funds — The carrying amounts of money market funds reported as cash and cash equivalents in the condensed balance sheets approximate their fair values due to their short-term nature. The fair values of money market funds are determined by Level 1 inputs utilizing quoted prices (unadjusted) in active markets for identical assets.
U.S. Treasury Bills — As of March 31, 2026 and December 31, 2025, the Company had short-term U.S. Treasury bills. Fair values of these securities are determined by Level 2 inputs utilizing quoted prices
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
(unadjusted) in active markets for similar assets. The following table presents information about the Company’s investments in held-to-maturity U.S. Treasury bills as of each reported date (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | As of March 31, 2026 |
| Balance Sheet Location | | Original Maturities | | Amortized
Cost | | | | | | Estimated
Fair Value |
| | | | | | | | | | |
| Investments | | between 3 and 12 months | | $ | 533,281 | | | | | | | $ | 533,211 | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | | As of December 31, 2025 |
| Balance Sheet Location | | Original Maturities | | Amortized
Cost | | | | | | Estimated
Fair Value |
| Cash and cash equivalents | | less than 3 months | | $ | 9,948 | | | | | | | $ | 9,948 | |
| Investments | | between 3 and 12 months | | 483,714 | | | | | | | 483,891 | |
| | | | | | | | | | |
| Total | | | | $ | 493,662 | | | | | | | $ | 493,839 | |
Note 4. Commitments and Contingencies
Tanabe License Agreement
In July 2020, the Company entered into an exclusive license agreement (the Tanabe License) with Tanabe Pharma Corporation (Tanabe) (formerly Mitsubishi Tanabe Pharma Corporation), pursuant to which Tanabe granted the Company an exclusive, worldwide, royalty-bearing, sublicensable license under Tanabe’s patent and other intellectual property rights to exploit products incorporating lorundrostat (formerly MT-4129) (Lorundrostat Product) for the prevention, treatment, diagnosis, detection, monitoring, or predisposition testing with respect to indications, diseases, and conditions in humans. Pursuant to the Tanabe License, the Company paid Tanabe a $1.0 million upfront fee and development milestone payments of $9.0 million in the aggregate. The Company has remaining obligations to pay Tanabe commercial milestone payments of up to $155.0 million in the aggregate upon first commercial sale and upon meeting certain annual sales targets, as well as additional commercial milestone payments of up to $10.0 million for a second indication. Additionally, the Company is obligated to pay Tanabe tiered royalties at percentages ranging from the mid-single digits to ten percent (10%) of aggregate net sales of each Lorundrostat Product on a Lorundrostat Product-by-Lorundrostat Product and country-by-country basis, until the later of (i) the expiration of the last-to-expire valid Tanabe patent claim covering a Lorundrostat Product, (ii) ten years from the first commercial sale of a Lorundrostat Product, or (iii) the expiration of regulatory exclusivity in such country. Such royalties are subject to reduction under specified conditions, including lack of patent coverage and generic competition.
The Company has no remaining development milestone obligations under the Tanabe License and did not incur any development or commercial expenses pursuant to the Tanabe License during the three months ended March 31, 2026 and 2025.
The Company is obligated to use commercially reasonable efforts to conduct and complete the development activities and to file for regulatory approval for at least one Lorundrostat Product in a major market country and consider in good faith developing at least one Lorundrostat Product in a non-major market country. If the Company elects to sublicense its rights under the Tanabe License to a third party with respect to exploitation of lorundrostat or any Lorundrostat Product in certain countries in Asia, the Company has agreed to negotiate such a sublicense first, for a specified period of time, with Tanabe, if Tanabe notifies the Company that it would like to obtain such a sublicense. The Company also agreed not to commercialize any competing product prior to three years following the first commercial sale of the first Lorundrostat Product in any country without Tanabe’s prior consent.
Unless terminated earlier, the Tanabe License will continue until the expiration of all of the Company’s royalty obligations to Tanabe. The Company may terminate the Tanabe License for any or no reason upon 90 or 180 days’ prior written notice to Tanabe, depending on whether the Lorundrostat Product has received
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
regulatory approval. Tanabe may terminate the Tanabe License if the Company or its affiliates or sublicensees initiate a challenge to the patent rights licensed to the Company by Tanabe. In addition, either party may terminate the Tanabe License in the event of an uncured material breach by or bankruptcy of the other party, subject to certain notice and cure periods, or upon the other party’s bankruptcy or insolvency.
Litigation
Liabilities for loss contingencies arising from claims, assessments, litigation, fines, penalties, and other sources are recorded when it is probable that a liability has been incurred and the amount can be reasonably estimated. From time to time, the Company may become involved in legal proceedings arising in the ordinary course of business. The Company was not subject to any material legal proceedings during the three months ended March 31, 2026 and 2025, and no material legal proceedings are currently pending or threatened.
Indemnification Agreements
In the ordinary course of business, the Company may provide indemnification of varying scope and terms to vendors, lessors, business partners, and other parties with respect to certain matters including, but not limited to, losses arising from breach of such agreements or intellectual property infringement claims made by third parties. In addition, the Company has entered into indemnification agreements with officers of the Company and members of its board of directors that will require the Company, among other things, to indemnify them against certain liabilities that may arise by reason of their status or service as officers or directors. The maximum potential amount of future payments the Company could be required to make under these indemnification agreements is, in many cases, unlimited. To date, the Company has not incurred any material costs as a result of such indemnifications. The Company is not aware of any claims under indemnification arrangements, and it has not accrued any material liabilities related to such obligations in its condensed financial statements as of March 31, 2026 and December 31, 2025.
Note 5. Accrued Liabilities
Accrued liabilities consisted of the following (in thousands):
| | | | | | | | | | | |
| |
| | | |
| March 31, | | December 31, |
| 2026 | | 2025 |
| | | |
| Research and development expenses | $ | 4,587 | | | $ | 3,460 | |
| Compensation and benefits | 2,312 | | | 5,540 | |
| Professional fees and other | 6,703 | | | 4,096 | |
| Total | $ | 13,602 | | | $ | 13,096 | |
Note 6. Capital Stock
As of March 31, 2026, the Company had reserved authorized shares of common stock for future issuance as follows:
| | | | | |
| March 31, |
| 2026 |
| Common stock options outstanding | 7,420,692 | |
| Shares available for grant under the 2023 Plan | 2,866,922 | |
| Shares available for grant under the ESPP | 2,067,084 | |
Restricted stock units outstanding | 702,948 | |
| Pre-funded warrants issued and outstanding | 549,755 | |
| Shares available for grant under the 2025 Inducement Plan | 539,748 | |
| Total | 14,147,149 | |
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
At-the-Market Equity Offering Sales Agreements
On November 10, 2025, the Company entered into an ATM Equity Offering Sales Agreement (the ATM Agreement) with BofA Securities, Inc., Evercore Group L.L.C., and Goldman Sachs & Co. LLC, pursuant to which the Company may sell shares of its common stock having an aggregate offering price of up to $300.0 million from time to time. During the three months ended March 31, 2026, the Company sold pursuant to the ATM Agreement an aggregate of 568,320 shares of common stock at a weighted-average price of $35.66 per share for aggregate net proceeds of approximately $20.2 million after deducting commissions and offering expenses. There were no sales of common stock pursuant to the ATM Agreement during the three months ended March 31, 2025. From inception of the ATM Agreement and through March 31, 2026, the Company sold an aggregate of 2,720,508 shares of common stock at a weighted-average price of $41.45 per share for aggregate net proceeds of approximately $112.4 million after deducting commissions and offering expenses. As of March 31, 2026, approximately $187.2 million of shares remained available for sale pursuant to the ATM Agreement.
On March 21, 2024, the Company entered into an ATM Equity Offering Sales Agreement (the Prior ATM Agreement) with BofA Securities, Inc. and Evercore Group L.L.C. Effective November 9, 2025, the Prior ATM Agreement was terminated in connection with the execution of the ATM Agreement. There were no sales of common stock pursuant to the Prior ATM Agreement during each of the three months ended March 31, 2026 and March 31, 2025. From inception of the Prior ATM Agreement and through its termination effective November 9, 2025, the Company sold an aggregate of 1,914,040 shares of common stock at a weighted-average price of $14.32 per share for aggregate net proceeds of approximately $27.3 million after deducting commissions and offering expenses.
Note 7. Stock-Based Compensation
2023 Incentive Award Plan
The Company grants equity awards under the 2023 Incentive Award Plan (the 2023 Plan). During the three months ended March 31, 2026, the Company began granting restricted stock units under the 2023 Plan.
2025 Employment Inducement Incentive Award Plan
The Company maintains the 2025 Employment Inducement Incentive Award Plan (the 2025 Inducement Plan), under which non‑qualified stock options and restricted stock units may be granted in connection with the commencement of employment. During the three months ended March 31, 2026, the Company began granting restricted stock units under the 2025 Inducement Plan.
2020 Equity Incentive Plan
The 2020 Equity Incentive Plan (the 2020 Plan) continues to govern the terms of outstanding awards, although no new awards are granted under the plan.
As of March 31, 2026, the Company had the following balances by plan:
| | | | | | | | | | | | | | | | | | | | | | | |
| Options
Outstanding | | Unvested
Restricted Stock Awards | | Unvested
Restricted Stock Units | | Shares Available
for Grant |
| 2023 Plan | 6,841,701 | | | — | | | 568,040 | | | 2,866,922 | |
2025 Inducement Plan | 325,344 | | | — | | | 134,908 | | | 539,748 | |
| 2020 Plan | 253,647 | | | 64,714 | | | — | | | — | |
| Total | 7,420,692 | | | 64,714 | | | 702,948 | | | 3,406,670 | |
Mineralys Therapeutics, Inc.
Notes to Condensed Financial Statements
(unaudited)
2023 Employee Stock Purchase Plan
The Company maintains the 2023 Employee Stock Purchase Plan (the ESPP) under which eligible employees may purchase shares of common stock through payroll deductions. As of March 31, 2026, the Company had 2,067,084 shares available for issuance, and 57,839 cumulative shares had been issued under the ESPP.
Total stock-based compensation expense recognized was allocated as follows (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended | | |
| March 31, | | |
| 2026 | | 2025 | | | | |
| Research and development | $ | 2,476 | | | $ | 1,628 | | | | | |
| General and administrative | 4,072 | | | 2,017 | | | | | |
Total | $ | 6,548 | | | $ | 3,645 | | | | | |
Item 2. Management's Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis and the unaudited interim financial statements included in this Quarterly Report on Form 10-Q (Quarterly Report) should be read in conjunction with the financial statements and notes thereto for the year ended December 31, 2025 and the related Management’s Discussion and Analysis of Financial Condition and Results of Operations, both of which are contained in the Annual Report on Form 10-K for the year ended December 31, 2025.
Special Note Regarding Forward-Looking Statements
This Quarterly Report contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the Securities Act), and Section 21E of the Securities Exchange Act of 1934, as amended (the Exchange Act). All statements other than statements of historical facts contained in this Quarterly Report, including statements regarding our future results of operations and financial position, business strategy, research and development plans, the anticipated timing, costs, design, and conduct of our ongoing and planned preclinical studies and planned clinical trials for lorundrostat and any future product candidates, the timing and likelihood of regulatory filings and approvals for lorundrostat (including the anticipated timing of any U.S. Food and Drug Administration’s (FDA) approval of our new drug application (NDA) that was submitted to the FDA for lorundrostat for the treatment of hypertension when used in combination with other antihypertensive drugs in December 2025) and any future product candidates, our ability to commercialize our product candidates, if approved, the potential to develop future product candidates, the potential benefits of strategic collaborations and our intent to enter into any strategic arrangements, the timing and likelihood of success, plans and objectives of management for future operations and future results of anticipated product development efforts, and the sufficiency of our cash, cash equivalents and investments to fund our operations, are forward-looking statements. These statements involve known and unknown risks, uncertainties, and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by the forward-looking statements, including the timing, volume and nature of feedback or requests from the FDA in connection with our NDA submission, macroeconomic trends and uncertainty with regard to high interest rates, elevated inflation, tariffs and other trade policies, geopolitical conflict, and the potential for a local and/or global economic recession. This Quarterly Report also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions, and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk.
In some cases, you can identify forward-looking statements by terms such as “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” or “would,” or the negative of these terms or other similar expressions. The forward-looking statements in this Quarterly Report are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition, and results of operations. These forward-looking statements speak only as of the date of this Quarterly Report and are subject to a number of risks, uncertainties, and assumptions, including, without limitation, the risk factors described in Part II, Item 1A, “Risk Factors” in this Quarterly Report, in Part I, Item 1A, “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025, and under a similar heading in any other periodic or current report we may file with the U.S. Securities and Exchange Commission (the SEC) in the future. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances, or otherwise. All forward-looking statements are qualified in their entirety by this cautionary statement, which is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. This Quarterly Report includes trademarks, trade names, and service marks that are the property of other organizations. Solely for convenience, trademarks and trade names referred to in this Quarterly Report appear without the ® and ™ symbols, but those references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights, or that the applicable owner will not assert its rights, to these trademarks and trade names.
Overview
We are a biopharmaceutical company focused on developing medicines to target hypertension and related comorbidities such as chronic kidney disease (CKD), obstructive sleep apnea (OSA), and other diseases driven by dysregulated aldosterone. Our initial product candidate, lorundrostat, is a proprietary, orally administered, highly selective aldosterone synthase inhibitor.
We have now completed six clinical trials of lorundrostat supporting its efficacy and safety profile while also validating aldosterone as an integral therapeutic target in uncontrolled hypertension (uHTN) and resistant hypertension (rHTN). The clinical program includes two pivotal, registrational trials, the Phase 3 Launch-HTN trial and Phase 2 Advance-HTN trial, which support the robust, durable, and clinically meaningful reductions in systolic blood pressure (BP) by lorundrostat. Lorundrostat was well tolerated in both trials with a favorable safety profile. We submitted our NDA to the FDA in December 2025 for lorundrostat for the treatment of hypertension in combination with other antihypertensive drugs. The FDA accepted the NDA submission and provided us with a Prescription Drug User Fee Act (PDUFA) target date of December 22, 2026 for lorundrostat.
In the United States, there are approximately 120 million patients with sustained elevated BP, or hypertension. Approximately 60 million patients are treated and over 30 million do not achieve their BP goal, with approximately 20 million having systolic BP levels greater than 140 mmHg. Patients with hypertension that persists despite taking two or more medications have 1.8 and 2.5 times greater mortality risk due to either cardiovascular disease or stroke, respectively. Dysregulated aldosterone levels are a key factor in uHTN or rHTN in approximately 30% of patients.
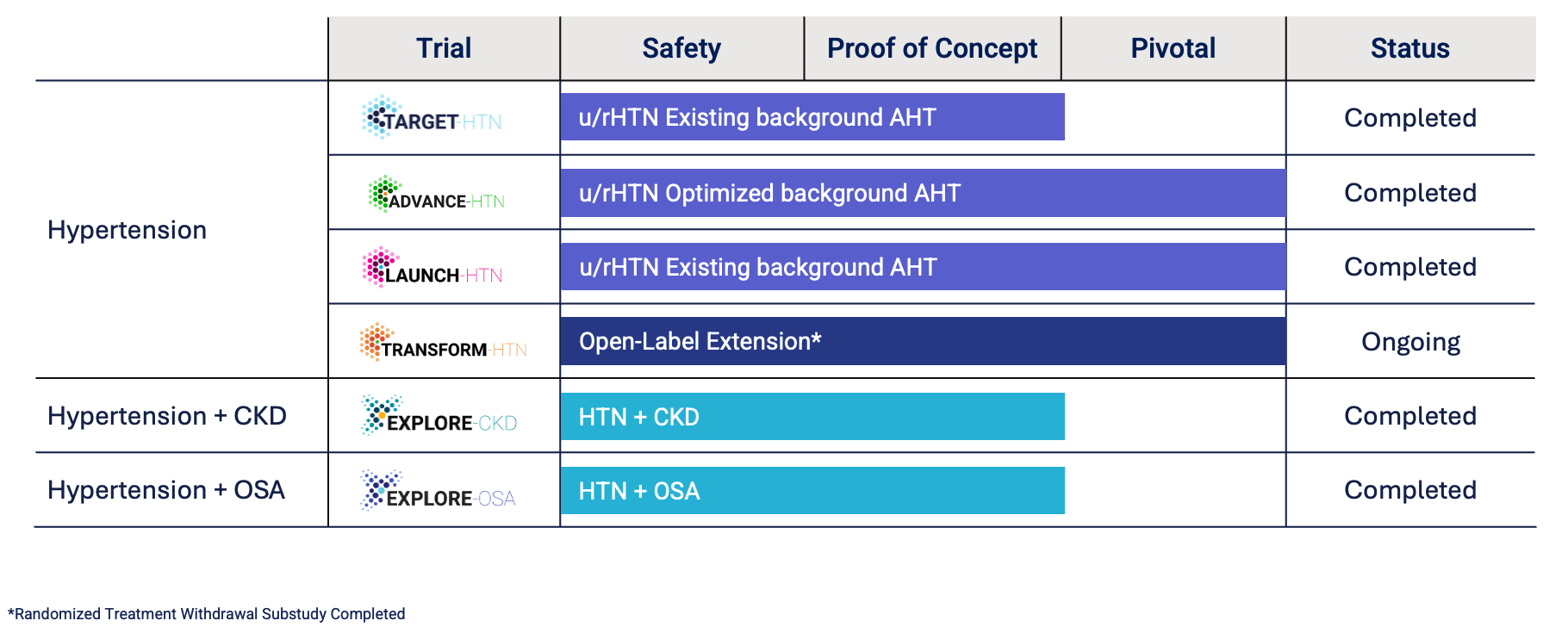
The image below summarizes the status of recently completed and ongoing clinical trials. Detailed results of our trials are set forth in Part I, Item 1, “Business” in our Annual Report on Form 10-K for the year ended December 31, 2025. Transform-HTN is an open-label extension trial that is ongoing and enables participants to continue to receive lorundrostat and allows us to gather additional long-term safety and efficacy data. All participants in the pivotal hypertension program, including the Advance-HTN and Launch-HTN trials, as well as the Explore-CKD trial, were given the opportunity to participate in the extension trial.
On March 9, 2026, we announced topline data from our exploratory Phase 2 Explore-OSA trial that evaluated the effect of lorundrostat in the treatment of overweight and obese participants with moderate-to-severe OSA and hypertension. After four weeks of treatment, lorundrostat 50 mg dosed in the evening did not demonstrate a clinically meaningful difference relative to placebo on the apnea-hypopnea index, the primary endpoint. The trial demonstrated a clinically meaningful reduction in BP at week four, with an 11.1 mmHg (p <
0.0001) and a 1.0 mmHg (p = NS) BP reduction with lorundrostat and placebo, respectively, in the pre-planned parallel arm analysis of the first period. There was a 6.2 mmHg placebo-adjusted reduction (p < 0.0003) in BP in the crossover analysis. Lorundrostat demonstrated a favorable safety profile and was well tolerated, with no serum potassium excursions above 5.5 mmol/L. Analysis is ongoing for other endpoints in the trial and may be reported in future publications or medical meetings. Detailed results of our trials are set forth in Part I, Item 1, “Business” in our Annual Report on Form 10-K for the year ended December 31, 2025. Financial Overview
We commenced our operations in May 2019 and have devoted substantially all of our resources to date to organizing and staffing our company, business planning, raising capital, in-licensing our product candidate, lorundrostat, establishing our intellectual property portfolio, conducting research, preclinical studies, and clinical trials, and providing other general and administrative support for our operations. As of March 31, 2026, we had cash, cash equivalents, and investments of $646.1 million. Since inception, we have raised aggregate gross proceeds of approximately $1.1 billion from the sale of common stock, convertible preferred stock, pre-funded warrants, and convertible notes. Our net losses for the three months ended March 31, 2026 and 2025 were $39.3 million and $42.2 million, respectively. As of March 31, 2026 and December 31, 2025, we had an accumulated deficit of $496.5 million and $457.2 million, respectively. Our net losses may fluctuate significantly from quarter to quarter and year to year, depending on the timing of our clinical development activities and other research and development activities.
We anticipate that certain expenses will increase substantially as we prepare for a potential approval and launch of lorundrostat, including the following:
•conducting ongoing regulatory activities, including responding to FDA information requests, supporting a potential advisory committee meeting (not anticipated currently), preparing for potential approval and post-marketing commitments, and commercial launch of lorundrostat;
•continuing to expand our pre-commercial organization, including building out medical affairs, market access, health economics and outcomes research, regulatory, quality, manufacturing, and commercial functions;
•obtaining, maintaining, protecting, and enforcing our intellectual property;
•continuing the Transform-HTN open-label extension trial and conducting any required post-approval studies;
•attracting and retaining experienced scientific, regulatory, medical, commercial, and operational talent; and
•operating as a public company with expanding SEC compliance, legal, and finance obligations.
We have never generated any revenue and do not expect to generate any revenue from product sales unless and until we obtain regulatory approval for lorundrostat, if ever. Accordingly, until such time as we can generate significant revenue from sales of lorundrostat, if ever, we expect to finance our cash needs through equity offerings, debt financings, or other capital sources, including potential collaborations, licenses, and other similar arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Our failure to raise capital or enter into such other arrangements when needed would have a negative impact on our financial condition and could force us to delay, limit, reduce, or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves. For more information, see “Liquidity and Capital Resources.”
Tanabe License Agreement
In July 2020, we entered into an exclusive license agreement (the Tanabe License) with Tanabe Pharma Corporation (Tanabe) (formerly Mitsubishi Tanabe Pharma Corporation), pursuant to which Tanabe granted us an exclusive, worldwide, royalty-bearing, sublicensable license under Tanabe’s patent and other intellectual property rights to exploit products incorporating lorundrostat (formerly MT-4129) (Lorundrostat Product) for the prevention, treatment, diagnosis, detection, monitoring, or predisposition testing with respect to indications, diseases, and conditions in humans. Pursuant to the Tanabe License, we paid Tanabe a $1.0 million upfront fee and development milestone payments of $9.0 million in the aggregate. We have remaining obligations to pay Tanabe commercial milestone payments of up to $155.0 million in the aggregate upon first commercial sale and upon meeting certain annual sales targets, as well as additional commercial milestone payments of up to $10.0 million for a second indication. Additionally, we are obligated to pay Tanabe tiered royalties at percentages ranging from the mid-single digits to ten percent (10%) of aggregate net sales of each Lorundrostat Product on a Lorundrostat Product-by-Lorundrostat Product and country-by-country basis, until the later of (i) the expiration of the last-to-expire valid Tanabe patent claim covering a Lorundrostat Product, (ii) ten years from the first commercial sale of a Lorundrostat Product, or (iii) the expiration of regulatory exclusivity in such country. Such royalties are subject to reduction under specified conditions, including lack of patent coverage and generic competition. We have no remaining development milestone obligations under the Tanabe License and did not incur any development or commercial expenses pursuant to the Tanabe License during the three months ended March 31, 2026 and 2025.
We are obligated to use commercially reasonable efforts to conduct and complete the development activities and to file for regulatory approval for at least one Lorundrostat Product in a major market country and consider in good faith developing at least one Lorundrostat Product in a non-major market country. If we elect to sublicense our rights under the Tanabe License to a third party with respect to exploitation of lorundrostat or any Lorundrostat Product in certain countries in Asia, we have agreed to negotiate such a sublicense first, for a specified period of time, with Tanabe, if Tanabe notifies us that it would like to obtain such a sublicense. We also agreed not to commercialize any competing product prior to three years following the first commercial sale of the first Lorundrostat Product in any country without Tanabe’s prior consent.
Public Offerings
On March 11, 2025, we entered into an underwriting agreement relating to the issuance and sale of 14,907,406 shares of our common stock at a price of $13.50 per share for net proceeds of approximately $188.7 million after deducting an underwriting discount of 6% and offering expenses. The offering was made pursuant to our shelf registration statement on Form S-3 (Registration Statement No. 333-278122) previously filed with and declared effective by the SEC (the Registration Statement), and a prospectus supplement and accompanying prospectus filed with the SEC. We are using the net proceeds from this offering to fund the clinical development of lorundrostat, including research and development, manufacturing, and pre-commercialization activities, as well as for working capital and general corporate purposes.
At-the-Market Equity Offering Sales Agreements
On November 10, 2025, we entered into an ATM Equity Offering Sales Agreement (the ATM Agreement) with BofA Securities, Inc., Evercore Group L.L.C., and Goldman Sachs & Co. LLC, pursuant to which we may sell shares of our common stock having an aggregate offering price of up to $300.0 million from time to time. During the three months ended March 31, 2026, we sold pursuant to the ATM Agreement an aggregate of 568,320 shares of common stock at a weighted-average price of $35.66 per share for aggregate net proceeds of approximately $20.2 million after deducting commissions and offering expenses. There were no sales of common stock pursuant to the ATM Agreement during the three months ended March 31, 2025. From inception of the ATM Agreement and through March 31, 2026, we sold an aggregate of 2,720,508 shares of common stock at a weighted-average price of $41.45 per share for aggregate net proceeds of approximately $112.4 million after deducting commissions and offering expenses. As of March 31, 2026, approximately $187.2 million of shares remained available for sale pursuant to the ATM Agreement.
On March 21, 2024, we entered into an ATM Equity Offering Sales Agreement (the Prior ATM Agreement) with BofA Securities, Inc. and Evercore Group L.L.C. Effective November 9, 2025, the Prior ATM Agreement was terminated in connection with the execution of the ATM Agreement. There were no sales of common stock pursuant to the Prior ATM Agreement during each of the three months ended March 31, 2026 and March 31, 2025. From inception of the Prior ATM Agreement and through its termination effective November 9, 2025, we sold an aggregate of 1,914,040 shares of common stock at a weighted-average price of $14.32 per share for aggregate net proceeds of approximately $27.3 million after deducting commissions and offering expenses.
Key Components of Results of Operations
Research and Development
Research and development expenses consist primarily of external and internal costs related to the development of lorundrostat. Research and development expenses are recognized as incurred, and payments made prior to the receipt of goods or services to be used in research and development are capitalized until the goods are received or the services are performed.
Research and development expenses include:
•compensation costs, including salaries, benefits, and stock-based compensation for our research and development personnel;
•external research and development expenses incurred under agreements with contract research organizations and consultants to conduct and support our clinical trials of lorundrostat;
•costs related to clinical supply, manufacturing, and regulatory activities;
•costs related to advancing our commercial readiness activities in preparation for a potential launch of lorundrostat for patients with hypertension, if approved by the FDA; and
•allocated overhead.
Our research and development expenses have been primarily driven by the timing and phase of our clinical trials, including the initiation and completion of studies, the number of trials in progress, and the size and complexity of each trial. We expect certain research and development expenses related to our clinical trial activities to decline in the upcoming periods as most trials have been completed relative to prior periods. However, we anticipate that certain other expenses will increase substantially as we:
•conduct ongoing regulatory activities, including responding to FDA information requests, supporting a potential advisory committee meeting (not anticipated currently), preparing for potential approval and post-marketing commitments, and commercial launch of lorundrostat;
•continue the Transform-HTN open-label extension trial and conduct any required post-approval studies; and
•attract and retain experienced scientific, regulatory, medical, commercial, and operational talent.
We cannot determine with certainty the timing of initiation, the duration, or the completion costs of current or future clinical trials and preclinical studies of lorundrostat or any future product candidates due to the inherently unpredictable nature of clinical and preclinical development. Clinical and preclinical development timelines, the probability of success, and development costs can differ materially from expectations. In addition, we cannot forecast whether lorundrostat or any future product candidates may be subject to future collaborations, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
Our future development costs may vary significantly based on factors such as:
•the initiation, type, number, scope, development phase, progress, duration, expansions, results, costs, and timing of clinical trials and preclinical studies of lorundrostat and any future product candidates we may choose to pursue, including any modifications to clinical development plans based on feedback that we may receive from regulatory authorities;
•our ability and strategic decision to develop future product candidates other than lorundrostat, and the timing of such development, if any;
•our ability to receive timely regulatory approvals for lorundrostat, any future product candidates, and any additional indications of lorundrostat and any future product candidates, in the jurisdictions in which we or any future partners apply for such approvals;
•the costs and timing of manufacturing lorundrostat or any future product candidates for use in our trials, including as a result of inflation, changes in international trade policies and tariffs, any supply chain issues, or component shortages;
•any additional jurisdictions in which we may seek approval for lorundrostat and any future product candidates and the timing of seeking approval in such jurisdictions; and
•the extent to which we establish strategic collaborations or other arrangements.
General and Administrative Expenses
General and administrative expenses consist primarily of (i) compensation costs, including salaries, benefits, and stock-based compensation, for executive and administrative personnel; (ii) professional fees for legal, audit, tax, and other consulting or advisory services; (iii) costs associated with building our pre-commercial organization, including medical affairs, market access, and health economics; (iv) fees relating to intellectual property and corporate matters; and (v) allocated overhead.
We expect general and administrative expenses to increase as we build out our pre-commercial and commercial capabilities in preparation for the potential approval and launch of lorundrostat; obtain, maintain, protect, and enforce our intellectual property; and attract and retain scientific, regulatory, medical, commercial, and operational talent, and as we incur higher costs associated with being a more mature public company.
Other Income, Net
Interest Income, Net
Interest income reported in each period is associated with our investments in money market funds and U.S. treasuries, net of fees, or other related expenses.
Comparison of the Three Months Ended March 31, 2026 and 2025
| | | | | | | | | | | | | | | | | |
| Three Months Ended, | | |
| March 31, | | |
| 2026 | | 2025 | | Change |
| (in thousands) |
| | | | | |
| | | | | |
| Research and development expenses | $ | (24,365) | | | $ | (37,879) | | | $ | 13,514 | |
| General and administrative expenses | (20,975) | | | (6,568) | | | (14,407) | |
| Total other income, net | 6,001 | | | 2,236 | | | 3,765 | |
| Net loss | $ | (39,339) | | | $ | (42,211) | | | $ | 2,872 | |
Research and Development Expenses
Research and development expenses decreased by $13.5 million for the three months ended March 31, 2026, compared to the three months ended March 31, 2025. The decrease was primarily driven by a $15.5 million reduction in preclinical and clinical costs following the conclusion of the lorundrostat pivotal program in the second quarter of 2025. This decrease was partially offset by $1.1 million of increased clinical supply, manufacturing, and regulatory costs, and $0.8 million of increased personnel-related expenses resulting from headcount growth and increased compensation.
General and Administrative Expenses
General and administrative expenses increased by $14.4 million for the three months ended March 31, 2026, compared to the three months ended March 31, 2025. The increase was primarily driven by $7.9 million of higher professional fees, $6.1 million of increased personnel-related expenses resulting from headcount growth and increased compensation, and $0.4 million of other general and administrative expenses.
Total Other Income, Net
Total other income, net increased by $3.8 million for the three months ended March 31, 2026, compared to the three months ended March 31, 2025. The increase reflects higher interest earned on investments in money market funds and U.S. Treasuries due to higher average cash balances invested during the three months ended March 31, 2026.
Liquidity and Capital Resources
We have incurred net losses and negative cash flows from operations since our inception and we expect to continue to incur significant expenses and operating losses for the foreseeable future. Since inception, we have raised aggregate gross proceeds of approximately $1.1 billion from the sale of common stock, convertible preferred stock, pre-funded warrants, and convertible notes. Our primary uses of cash to date have been to fund our research and development and other activities, including with respect to lorundrostat, business planning, establishing and maintaining our intellectual property portfolio, hiring personnel, raising capital, and providing general and administrative support for these operations.
As of March 31, 2026, we had cash, cash equivalents, and investments of $646.1 million and an accumulated deficit of $496.5 million. During the three months ended March 31, 2026, we sold an aggregate of 568,320 shares of common stock under the ATM Agreement at a weighted-average price of $35.66 per share for aggregate net proceeds of approximately $20.2 million after deducting commissions and offering expenses. As of March 31, 2026, approximately $187.2 million of shares remained available for sale pursuant to the ATM Agreement, subject to the terms and conditions of the ATM Agreement and applicable securities laws.
Funding Requirements
Based on our current operating plan, we believe that our cash, cash equivalents, and investments as of March 31, 2026 will be sufficient to allow us to fund our operations for at least twelve months. However, our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement that involves risks and uncertainties, and actual results could vary materially. We have based this estimate on assumptions that may prove to be wrong, and we could deplete our capital resources sooner than we expect. Additionally, the process of testing product candidates in clinical trials is costly, and the timing of progress and expenses in these trials is uncertain.
Our future capital requirements will depend on many factors, including, but not limited to:
•the initiation, type, number, scope, development phase, progress, duration, expansions, results, costs, and timing of clinical trials and preclinical studies of lorundrostat and any future product
candidates we may choose to pursue, including any modifications to clinical development plans based on feedback that we may receive from regulatory authorities;
•our ability and strategic decision to develop future product candidates other than lorundrostat, and the timing of such development, if any;
•our ability to receive timely regulatory approvals for lorundrostat, any future product candidates, and additional indications of lorundrostat and any future product candidates, in the jurisdictions in which we or any future partners apply for such approvals;
•the costs and timing of manufacturing for lorundrostat, or any future product candidate, including commercial manufacture at sufficient scale, if any product candidate is approved, including as a result of inflation, changes in international trade policies and tariffs, any supply chain issues, or component shortages;
•any additional jurisdictions in which we may seek approval for lorundrostat and any future product candidates and timing of seeking approval in such jurisdictions;
•the costs, timing, and outcome of regulatory approval of lorundrostat or any future product candidates;
•the costs of obtaining, maintaining, enforcing, and protecting our patents and other intellectual property and proprietary rights;
•our efforts to enhance operational systems and hire additional personnel to satisfy our obligations as a public company, including enhanced internal control over financial reporting;
•the costs associated with hiring additional personnel and consultants as our business grows, including additional executive officers and clinical development, regulatory, manufacturing, quality, and commercial personnel;
•the timing and amount of the milestone, royalty, or other payments we must make to Tanabe, from whom we have in-licensed lorundrostat, or any future licensors;
•the costs and timing of establishing or securing sales and marketing capabilities if lorundrostat or any future product candidate is approved;
•our ability to achieve sufficient market acceptance, coverage, and adequate reimbursement from third-party payors, and adequate market share and revenue for any approved products;
•patients’ willingness to pay out-of-pocket for any approved products in the absence of coverage and/or adequate reimbursement from third-party payors;
•the terms and timing of establishing and maintaining collaborations, licenses, and other similar arrangements;
•costs associated with any products or technologies that we may in-license or acquire;
•any delays and cost increases that may result from any pandemic or other healthcare emergency; and
Until such time, if ever, as we can generate substantial product revenue, we expect to finance our cash needs through equity offerings, debt financings, or other capital sources, including potential collaborations,
licenses, and other similar arrangements. We do not have any committed external source of funds. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our stockholders may be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our common stockholders. Debt financing and equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures, or declaring dividends. Our ability to raise additional funds may be adversely impacted by potential worsening global economic conditions and the disruptions to, and volatility in, the credit and financial markets in the United States and worldwide resulting from factors that include, but are not limited to, geopolitical conflict in and around Ukraine, Israel, Iran, Venezuela, and other areas of the world, inflation, changes in international trade policies and tariffs, diminished liquidity and credit availability, declines in consumer confidence, declines in economic growth, increases in unemployment rates, and uncertainty about economic stability. If the equity and credit markets deteriorate, it may make any necessary debt or equity financing more difficult, more costly, and more dilutive. If we raise additional funds through future collaborations, licenses, or other similar arrangements with third parties, we may have to relinquish valuable rights to our future revenue streams, product candidates, research programs, intellectual property or proprietary technology, or grant licenses on terms that may not be favorable to us and/or may reduce the value of our common stock. If we are unable to raise additional funds through equity or debt financings or other arrangements when needed or on terms acceptable to us, we may be required to delay, limit, reduce, or terminate our product development or future commercialization efforts or grant rights to develop and market our product candidates even if we would otherwise prefer to develop and market such product candidates ourselves, or on less favorable terms than we would otherwise choose.
Cash Flows
Comparison of the Three Months Ended March 31, 2026 and 2025
Since our inception, we have primarily used our available cash to fund expenditures related to the in-license and development of lorundrostat. The following table sets forth a summary of cash flows for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Three Months Ended | | |
| March 31, | | |
| 2026 | | 2025 | | Change |
| |
| Net cash provided by (used in): | | | | | |
| Operating activities | $ | (39,471) | | | $ | (45,487) | | | $ | 6,016 | |
| Investing activities | (44,943) | | | (92,876) | | | 47,933 | |
| Financing activities | 24,272 | | | 189,279 | | | (165,007) | |
| Net | $ | (60,142) | | | $ | 50,916 | | | $ | (111,058) | |
Operating Activities
Net cash used in operating activities was $39.5 million during the three months ended March 31, 2026, compared to $45.5 million during the three months ended March 31, 2025, resulting in a decrease that was primarily attributable to a decrease in cash used to support our operating activities, including, but not limited to, the development of lorundrostat and related clinical trial expenses, personnel and compensation expense, legal and professional fees to support our operations, and general working capital requirements. The $6.0 million decrease in cash used consisted of the net effect of a decrease in net loss, adjusted for non-cash expenses, of approximately $2.2 million and changes in working capital of $3.8 million.
Investing Activities
Net cash used in investing activities was $44.9 million for the three months ended March 31, 2026, compared to $92.9 million for the three months ended March 31, 2025. Net cash used in investing activities
during the three months ended March 31, 2026 and 2025 was primarily driven by the timing and volume of maturities and purchases of marketable securities in each period. During the three months ended March 31, 2026, there was a $267.5 million increase in maturities of previously purchased marketable securities and a $219.6 million increase in purchases of marketable securities, compared to the three months ended March 31, 2025.
Financing Activities
Net cash provided by financing activities was $24.3 million during the three months ended March 31, 2026, compared to $189.3 million during the three months ended March 31, 2025. During the three months ended March 31, 2026, we received net proceeds of $20.2 million from the sale of common stock pursuant to the ATM Agreement, net of issuance costs, and during the three months ended March 31, 2025, we received net proceeds of $189.2 million from the sale of common stock in a public offering, net of offering costs. Additionally, during the three months ended March 31, 2026, we received increased proceeds of $3.9 million from stock option exercises as compared to the three months ended March 31, 2025.
Contractual Obligations and Commitments
Under the Tanabe License, we have milestone payment obligations that are contingent upon the achievement of specified levels of product sales and are required to make certain royalty payments in connection with the sale of products developed under the agreement. We are currently unable to estimate the timing or likelihood of achieving other future milestones or making future product sales. See above and Note 4. “Commitments and Contingencies” to our condensed financial statements included elsewhere in this Quarterly Report for additional information regarding the Tanabe License.
We enter into contracts in the normal course of business for contract research services, contract manufacturing services, professional services, and other services and products for operating purposes. These contracts generally provide for termination after a notice period, and, therefore, are cancelable contracts.
Critical Accounting Estimates
We have prepared the condensed financial statements in accordance with accounting principles generally accepted in the United States. The preparation of these condensed financial statements requires us to make estimates, assumptions, and judgments that affect the reported amounts of assets, liabilities, expenses, and related disclosures at the date of the condensed financial statements, and the reported amounts of expenses during the reporting period. On an ongoing basis, management evaluates its critical estimates, including those related to prepaid and accrued research and development expenses. We base our estimates on our historical experience and on assumptions that we believe are reasonable; however, actual results may differ materially from these estimates under different assumptions or conditions.
There were no changes during the three months ended March 31, 2026 to our critical accounting estimates as disclosed in our Annual Report on Form 10-K for the year ended December 31, 2025. For information on our significant accounting policies, please refer to Note 2. “Summary of Significant Accounting Policies” within our Annual Report on Form 10-K for the year ended December 31, 2025.
JOBS Act and Smaller Reporting Company Status
As an emerging growth company under the Jumpstart Our Business Startups Act of 2012, as amended (the JOBS Act), we can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to use the extended transition period for complying with new or revised accounting standards, and as a result of this election, our financial statements may not be comparable to those of companies that comply with public company effective dates. We intend to rely on other exemptions provided by the JOBS Act, including, without
limitation, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act of 2002.
We will remain an emerging growth company until the earliest of (i) the last day of the fiscal year following the fifth anniversary of the consummation of our initial public offering in February 2023, (ii) the last day of the fiscal year in which we have total annual gross revenue of at least $1.235 billion, (iii) the last day of the fiscal year in which we are deemed to be a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if, among other factors, the market value of our common stock held by non-affiliates exceeded $700.0 million as of the last business day of the second fiscal quarter of such year (subject to certain conditions), or (iv) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period.
We are also a smaller reporting company as defined in the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
Recently Issued Accounting Pronouncements
We have reviewed all recently issued accounting pronouncements and have determined that, other than as disclosed elsewhere in this Quarterly Report, such standards do not have a material impact on our condensed financial statements or do not otherwise apply to our operations.
Item 3. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
We are exposed to market risk related to changes in interest rates of our investment portfolio of cash equivalents and investments. As of March 31, 2026, our cash equivalents and investments consisted of money market funds and U.S. treasury securities. Our primary exposure to market risk is interest income sensitivity, which is affected by changes in the general level of U.S. interest rates. The fair value of our short-term cash equivalents and investments is subject to change as a result of potential changes in market interest rates. Due to the nature of our cash equivalents and investments, we believe an immediate hypothetical 10% change in interest rates would not have had a material effect on our results of operations during the periods presented.
Foreign Currency Exchange Risk
We are exposed to market risk related to changes in foreign currency exchange rates. We contract with vendors that are located outside the United States and certain invoices are denominated in foreign currencies. We are subject to fluctuations in foreign currency rates in connection with these arrangements. To date, these fluctuations have not been significant, and we have not had a formal hedging program with respect to foreign currency. We believe an immediate hypothetical 10% change in exchange rates would not have had a material effect on our results of operations during the periods presented.
Effects of Inflation
Inflation generally affects us by increasing our cost of labor and research and development contract costs. Although we do not believe that inflation has had a material impact on our financial position or results of operations to date, we may experience some effect in the future due to an impact on the costs to conduct clinical trials, labor costs we incur to attract and retain qualified personnel, and other operational costs. Inflationary costs could adversely affect our business, financial condition, and results of operations.
Item 4. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our periodic and current reports that we file with the SEC is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate, to allow timely decisions regarding required disclosures. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable and not absolute assurance of achieving the desired control objectives. In reaching a reasonable level of assurance, management was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures. In addition, the design of any system of controls is also based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Over time, control may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the design and operation of our disclosure controls and procedures, as such term is defined in Rules 13a-15(e) and 15d-15(e) promulgated under the Exchange Act, as of the end of the period covered by this Quarterly Report. Based on that evaluation, our principal executive officer and principal financial officer concluded that our disclosure controls and procedures were effective and were operating at a reasonable assurance level as of March 31, 2026.
Changes in Internal Control over Financial Reporting
Management has determined that there were no changes in our internal control over financial reporting that occurred during the three months ended March 31, 2026 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Part II - Other Information
Item 1. Legal Proceedings
We are not currently a party to any material legal proceeding. From time to time, we may become involved in legal proceedings arising in the ordinary course of our business. Regardless of outcome, litigation can have an adverse impact on us due to defense and settlement costs, diversion of management resources, negative publicity, reputational harm, and other factors.
Item 1A. Risk Factors
Our business, financial condition, and operating results may be affected by a number of factors, whether currently known or unknown, including, but not limited to, those described in Part I, Item 1A, “Risk Factors” of our Annual Report on Form 10-K for the year ended December 31, 2025. Any one or more of such factors could directly or indirectly cause our actual results of operations and financial condition to vary materially from past or anticipated future results of operations and financial condition. Any of these factors, in whole or in part, alone or combined with any of the other factors, could materially and adversely affect our business, financial condition, results of operations, and stock price. There have been no material changes to our risk factors since our Annual Report on Form 10-K for the year ended December 31, 2025. Item 2. Unregistered Sales of Equity Securities and Use of Proceeds
None.
Item 3. Defaults Upon Senior Securities
Not Applicable.
Item 4. Mine Safety Disclosures
Not Applicable.
Item 5. Other Information
During the three months ended March 31, 2026, an affiliate of the Company, Catalys Pacific Fund, LP, entered into a Rule 10b5-1 trading plan. Catalys Pacific Fund GP, LP is the general partner of Catalys Pacific Fund, LP. Brian Taylor Slingsby, M.D., Ph.D., M.P.H., one of our directors, is the managing director of Catalys Pacific, LLC, the general partner of Catalys Pacific Fund GP, LP, and has voting and investment power over the shares held by Catalys Pacific Fund, LP. The material terms of this Rule 10b5-1 trading arrangement are described below:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name and Title | | Action Taken | | Type of Trading Arrangement | | Nature of Trading Arrangement | | Duration of Trading Arrangement | | Number of Securities |
| | | | | | | | | | |
Catalys Pacific Fund, LP | | Adoption January 9, 2026 | | Trading plan intended to satisfy the affirmative defense conditions of Exchange Act Rule 10b5-1(c) | | Sale of the Company’s common stock pursuant to the terms of the plan | | May 9, 2026 to March 31, 2027 | | 2,000,000 |
Except as set forth above, during the three months ended March 31, 2026, none of our directors or officers (as defined in Rule 16a-1(f) of the Exchange Act) adopted or terminated any contract, instruction, or written plan for the purchase or sale of our securities that was intended to satisfy the affirmative defense conditions of Rule 10b5-1(c) of the Exchange Act or any non-Rule 10b5-1 trading arrangement (as defined in the SEC’s rules).
Item 6. Exhibits
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Exhibit Number | | | Incorporated by Reference | Filed Herewith | | | | | | | | | | |
| Exhibit Description | Form | Date | Number | | | | | | | | | | |
| 3.1 | | | 8-K | 2/14/23 | 3.1 | | | | | | | | | | | |
| 3.2 | | | 8-K | 2/14/23 | 3.2 | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
10.1# | | | | | | x | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| 31.1 | | | | | | x | | | | | | | | | | |
| 31.2 | | | | | | x | | | | | | | | | | |
| 32.1* | | | | | | x | | | | | | | | | | |
| 32.2* | | | | | | x | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| 101.INS | | XBRL Instance Document | | | | x | | | | | | | | | | |
| 101.SCH | | XBRL Taxonomy Extension Schema Document | | | | x | | | | | | | | | | |
| 101.CAL | | XBRL Taxonomy Extension Calculation Linkbase Document | | | | x | | | | | | | | | | |
| 101.DEF | | XBRL Taxonomy Extension Definition Linkbase Document | | | | x | | | | | | | | | | |
| 101.LAB | | XBRL Taxonomy Extension Label Linkbase Document | | | | x | | | | | | | | | | |
| 101.PRE | | XBRL Taxonomy Extension Presentation Linkbase Document | | | | x | | | | | | | | | | |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | | | | x | | | | | | | | | | |
__________________
# Indicates management contract or compensatory plan.
* This certification is deemed not filed for the purpose of Section 18 of the Exchange Act or otherwise subject to the liability of that section, nor shall it be deemed incorporated by reference into any filing under the Securities Act or the Exchange Act.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | | | | |
| | MINERALYS THERAPEUTICS, INC. |
| | | |
| | | |
| Date: | May 6, 2026 | By: | /s/ Jon Congleton |
| | | Jon Congleton |
| | | President and Chief Executive Officer |
| | | (Principal Executive Officer) |
| | | |
| Date: | May 6, 2026 | By: | /s/ Adam Levy |
| | | Adam Levy |
| | | Chief Financial Officer and Secretary |
| | | (Principal Financial Officer; Principal Accounting Officer) |